Botulinum Neurotoxins for Postã¢â‚¬âstroke Spasticity in Adults a Systematic Review
- Study protocol
- Open Access
- Published:
Report design and methods of the BoTULS trial: a randomised controlled trial to evaluate the clinical effect and cost effectiveness of treating upper limb spasticity due to stroke with botulinum toxin type A
Trials volume 9, Article number:59 (2008) Cite this commodity
Abstract
Groundwork
Following a stroke, 55–75% of patients feel upper limb bug in the longer term. Upper limb spasticity may cause hurting, deformity and reduced function, affecting mood and independence. Botulinum toxin is used increasingly to treat focal spasticity, just its affect on upper limb part after stroke is unclear.
The aim of this study is to evaluate the clinical and cost effectiveness of botulinum toxin type A plus an upper limb therapy programme in the handling of post stroke upper limb spasticity.
Methods
Trial pattern : A multi-centre open label parallel grouping randomised controlled trial and economic evaluation.
Participants : Adults with upper limb spasticity at the shoulder, elbow, wrist or mitt and reduced upper limb function due to stroke more than 1 month previously.
Interventions : Botulinum toxin type A plus upper limb therapy (intervention grouping) or upper limb therapy alone (control grouping).
Outcomes : Outcome assessments are undertaken at ane, three and 12 months. The primary outcome is upper limb office one month afterward study entry measured past the Action Research Arm Test (ARAT). Secondary outcomes include: spasticity (Modified Ashworth Calibration); grip strength; dexterity (Nine Hole Peg Exam); inability (Barthel Activities of Daily Living Index); quality of life (Stroke Touch on Scale, Euroqol EQ-5D) and attainment of patient-selected goals (Canadian Occupational Performance Measure). Health and social services resources use, adverse events, use of other antispasticity treatments and patient views on the treatment will be compared. Participants are clinically reassessed at 3, 6 and 9 months to determine the demand for repeat botulinum toxin type A and/or therapy.
Randomisation : A spider web based fundamental independent randomisation service.
Blinding : Upshot assessments are undertaken by an assessor who is blinded to the randomisation group.
Sample size : 332 participants provide 80% power to detect a 15% difference in handling successes between intervention and control groups. Treatment success is divers every bit improvement of 3 points for those with a baseline ARAT of 0–3 and 6 points for those with ARAT of 4–56.
Trial registration
ISRCTN78533119
EudraCT 2004-002427-forty
CTA 17136/0230/001
Funding
National Institute for Health Research, Health Technology Cess Plan.
Ipsen Ltd provide botulinum toxin blazon A (Dysport®).
Background
Upper limb spasticity following stroke
Upper limb impairment affects 85% of stroke patients, many of whom still feel issues in the longer term[1, 2]. Spasticity can occur following stroke and is challenging to define and measure. Most recently it has been described as "matted sensori-motor control, resulting from an upper motor neurone lesion, presenting equally intermittent or sustained involuntary activation of muscles"[three]. An older, narrower and more commonly quoted definition is " a motor disorder characterised past a velocity dependent increase in tonic stretch reflexes with exaggerated tendon reflexes, resulting from hyperexcitability of the stretch reflex"[4]. Withal, put simply spasticity is over activity of muscles as a result of harm to the brain or spinal cord. Spasticity can cause pain and deformity, and in the longer term may lead to the development of contractures[5, vi]. Upper limb spasticity tin pb to reduced arm function and bug with ease of hygiene[half dozen]. Effective management of spasticity requires a coordinated multidisciplinary team approach[6].
Randomised controlled trials of botulinum toxin in the handling of upper limb spasticity following stroke
Afterwards injection botulinum toxin causes local paresis of muscles every bit a result of blocking cholinergic transmission at the neuromuscular junction. The clinical treatment upshot lasts for three–4 months[six]. To date, nine randomised controlled trials have evaluated its employ for the treatment of upper limb spasticity following stroke [7–15], and three systematic reviews accept been published [16–xviii] (two trials[14, fifteen] and two systematic reviews were published following the start of this written report[17, 18]).
Trials have reported a measurable reduction in resistance to passive movement on the Modified Ashworth Scale within 6 weeks, which and then reduces towards 12–xvi weeks, oftentimes losing statistical significance at this time. The primary benefits of spasticity reduction announced to be in terms of global patient/physician ratings and itemised passive disability scores (notably hand hygiene). Merely one study (published after this study commenced) has reported an improvement in agile upper limb office[fifteen].
As the treatment outcome of botulinum toxin lasts only 3–4 months, injections demand to exist repeated to offering sustained benefit. Only one trial (published subsequently this report commenced) has considered the impact of echo injections[fourteen]. This has provided limited evidence to support continued apply of botulinum toxin for spasticity reduction.
Guidelines for the use of botulinum toxin in the handling of spasticity recommend that it should be used in combination with a rehabilitation programme[6], but no trial to date has attempted to standardise upper limb therapy. No study has looked at the cost-effectiveness of treatment. Although transient muscle weakness at higher doses of botulinum toxin is well recognised, studies reported no unexpected adverse events, however, the event reporting system was oft unclear.
Participants in previous studies were significantly younger (average historic period 52–66 years) than typical stroke patients (the average incident age of stroke is 75 years) and the studies were often undertaken in specialist rehabilitation centres. Multidisciplinary care on a stroke unit is currently the gold standard for stroke rehabilitation and benefits are seen regardless of age or stroke severity[19]. Evaluations of botulinum toxin should recruit participants of all ages from stroke services rather than tertiary referral centres to avoid selection bias and ensure results are applicative to routine intendance.
The aim of this report is to evaluate the clinical and price effectiveness of botulinum toxin type A plus an upper limb therapy programme in the treatment of mail service stroke upper limb spasticity.
Methods
Study design
This is a multi-centre open up-label parallel group randomised controlled trial comparison the clinical and cost effectiveness of botulinum toxin blazon A plus an upper limb therapy program with the upper limb therapy programme alone for the treatment of upper limb spasticity due to stroke in adults. Figure ane outlines the study method.
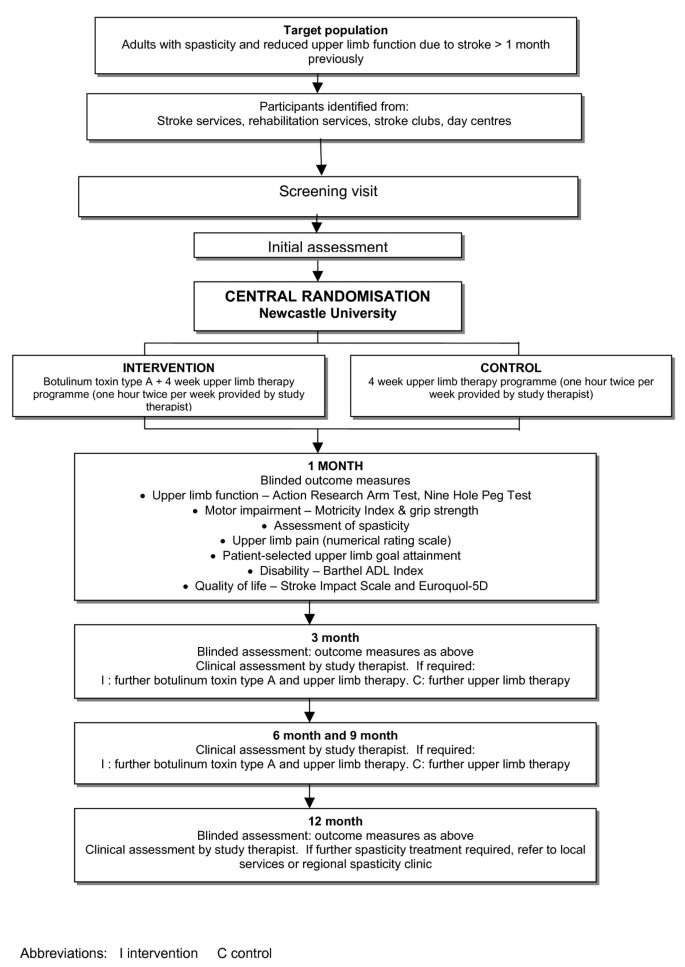
Study method.
Main objective
- 1.
To compare the upper limb office of participants with spasticity due to stroke who receive botulinum toxin type A injection(south) to the upper arm and/or forearm flexors/hand/shoulder girdle plus a iv week testify based upper limb therapy programme (intervention group) with participants who receive the upper limb therapy programme alone (control group) one month after study entry. Upper limb role is assessed using the Activity Research Arm Test (ARAT)[20].
Secondary objectives
- 1.
To compare the upper limb role and impairment of participants with spasticity due to stroke who receive botulinum toxin type A injection(s) to the upper arm and/or forearm flexors/hand/shoulder girdle plus a 4 week prove based upper limb therapy programme (intervention grouping) with participants who receive the upper limb therapy plan (control group) 1, 3 and 12 months afterwards study entry. Upper limb office and impairment is assessed by: ARAT[20], Motricity Index[21], grip strength[22], nine pigsty peg test[23], and Modified Ashworth calibration[24].
- 2.
To compare attainment of participant-selected upper limb goals, disability and stroke related quality of life between intervention and control groups at 1,3 and 12 months. The following measures are used: attainment of participant-selected upper limb goals (1 month only) – Canadian Occupational Functioning Measure (COPM)[25]; disability – Barthel ADL Alphabetize[26]; quality of life – Stroke Impact Scale[27], Euroqol EQ-5D[28], numerical rating scales for upper limb pain[29].
- three.
To seek the experience and views of participants well-nigh treatment at 1 and 12 months.
- 4.
To compare the health and social services resources used by control and intervention groups during the 12 months following study entry.
- 5.
To report adverse events and compare the use of other antispasticity treatments between intervention and command groups.
- 6.
To investigate the influence of severity of upper limb impairment and fourth dimension since stroke upon the efficacy of the intervention.
Setting
The study involves a collaborative network of twelve stroke services in the northward of England, with expertise in the direction of spasticity and apply of botulinum toxin being provided past the International Centre for Neurorehabilitation, Newcastle. Nosotros believe that the model which nosotros have developed i.e. stroke units with close links to a specialist spasticity service enables all stroke patients to access specialist care (both in terms of stroke and spasticity management) and this model could exist replicated in other settings.
Case ascertainment
- 1.
Potential participants are identified from a number of sources in each study heart (stroke unit, out-patients, day hospital and community rehabilitation teams). He/she is given an information leaflet and has an opportunity to discuss the study with a fellow member of the clinical team (grooming is given to clinical teams about the project and research governance). The research squad and then arranges to see him/her to talk over the study and seek consent at a screening visit.
- two.
There are potential participants who are non currently in contact with rehabilitation or stroke services. Local community stroke clubs and day centres accept been given information about the study and individuals may contact the report directly.
Inclusion criteria
Adults with a stroke greater than 1 month previously who have moderate/severe spasticity and reduced upper limb role who fulfil all of following criteria are eligible:
▪ Age over xviii years.
▪ At least 1 calendar month since stroke.
▪ Upper limb spasticity (Modified Ashworth Calibration[24] > two at the elbow and/or spasticity at the hand, wrist, or shoulder (there is no validated measure of spasticity at these sites)).
▪ Reduced upper limb function (ARAT[20] score 0–56)
▪ Able to comply with the requirements of the protocol and upper limb therapy programme.
▪ Informed consent given by participant or legal representative.
Exclusion criteria
▪ Significant speech or cognitive impairment which impedes ability to perform the ARAT[xx] assessment.
▪ Other significant upper limb impairment e.thou. fracture or frozen shoulder inside six months, astringent arthritis, amputation.
▪ Bear witness of fixed contracture.
▪ Pregnancy or lactating.
▪ Female at risk of pregnancy and not willing to take adequate precautions against pregnancy for the duration of the study.
▪ Other diagnosis likely to interfere with rehabilitation or outcome assessments eastward.g. registered bullheaded, malignancy.
▪ Other diagnosis which may contribute to upper limb spasticity east.g. multiple sclerosis, cerebral palsy.
▪ Contraindications to intramuscular injection.
▪ Religious objections to blood products (botulinum toxin type A (Dysport®) contains human being albumin).
▪ Contraindications to botulinum toxin type A which include bleeding disorders, myasthenia gravis and concurrent apply of aminoglycosides.
▪ Use of botulinum toxin to the upper limb in the previous three months.
▪ Known allergy or hypersensitivity to any of the examination compounds.
▪ Previous enrolment in this study.
Screening visit
Having sought consent, the screening cess is completed by a written report therapist or clinical research associate. The assessment consists of demographic details, review of medical history and medication; handedness; Abbreviated Mental Exam Score[30], Sheffield Aphasia Screening Exam[31], pre-stroke office (Oxford Handicap Scale)[32]; time since stroke; stroke type and subtype [33]; cocky reported electric current neurological impairment and office (Barthel ADL Alphabetize[26]); quality of life (Euroqol EQ-5D[28]). Details of current and anti-spasticity treatment received within the previous three months and concomitant medications are recorded.
Baseline assessment
The baseline visit is undertaken inside 2 weeks of the screening visit by a written report therapist or clinical research associate. The inclusion/exclusion criteria are reviewed to ensure that the participant is however eligible. Participants undergo a clinical assessment and are asked to consummate a bombardment of assessments including: Action Inquiry Arm Exam[20]; Motricity Index[21]; grip strength[22]; Nine Pigsty Peg Exam[23]; Modified Ashworth Scale[24]; upper limb pain[29] and Stroke Impact Scale[27]. Patient selected upper limb goals are besides identified (Canadian Occupational Operation Measure)[25]. Female participants of child-bearing potential (i.east. those who are not either surgically sterile or at least 1 twelvemonth post-last menstrual menstruum) have a urine pregnancy test, the result of which must be negative for the participant to be included in the report. Such participants must concord to use adequate contraception throughout the study if they are randomised to receive botulinum toxin type A. Participants are randomised one time the baseline assessment has been completed.
Randomisation
Randomisation is past a cardinal independent web based randomisation service from the Clinical Trials Unit, Newcastle University. Participants are stratified co-ordinate to level of upper limb role (ARAT 0–3, ARAT 4–28, ARAT 29–56) and randomised to intervention or control in a 1:1 ratio.
Botulinum toxin
Participants in the intervention grouping receive botulinum toxin type A (Dysport®). The range of muscles and dosages injected are described in 'The management of adults with spasticity using botulinum toxin: a guide to clinical practise'[6]. The maximum dose of botulinum toxin type A (Dysport®) administered at any one time indicate is 1000 units. All injectors are clinicians trained in the assessment and injection of botulinum toxin in the context of upper limb spasticity.
If further treatment is necessary at 3, six or 9 months a further injection is provided to those in the intervention group. At the 12-month review individuals in both the intervention and control groups who crave botulinum toxin are referred to the spasticity dispensary.
If during the course of the trial the study therapist decides that a participant in the control grouping has an unacceptable degree of symptomatic spasticity he/she discusses further direction with their stroke dr., physiotherapist, occupational therapist and/or a member of the local or regional spasticity team and the participant may and then be referred to the spasticity service for botulinum toxin.
The upper limb rehabilitation programme
Guidelines highlight that it is important that botulinum toxin is non used in isolation but as part of a comprehensive upper limb therapy plan[5, 6, 34]. Focal reductions in upper limb spasticity from any pharmacological intervention are unlikely to translate into sustained improvements in function or patient-selected rehabilitation goals without a targeted therapy plan.
The upper limb therapy programme is based upon available enquiry evidence from the stroke rehabilitation and skill acquisition literature as well equally clinical exercise [34–52], and consists of two menus. Participants with ARAT 0–3 receive bill of fare 1 which is designed specifically for participants with no active hand function and focuses on stretching, passive and active-assisted upper limb movement along with hand hygiene and positioning [42–47]. Bill of fare two is for participants with some retained agile upper limb motion (ARAT 4–56) and has been piloted in a previous written report[53]. Post-obit stretching of soft tissues affected past spasticity, this menu specifically concentrates on intensive task-orientated practise aimed at patient-centred goals. Upper limb goals are measured past the Canadian Occupational Performance Measure[25]. Each menu standardises the category of tasks, the number and order of repetitions as well as the corporeality of feedback for each session, simply within these parameters the therapist can tailor the specifics of each activity to the ability of the patient. Manuals and training programmes accept been adult for both upper limb therapy menus.
The upper limb therapy programme is provided by report therapists and each participant receives 1 hour per mean solar day, two times per week for four weeks, in addition to their other rehabilitation needs. The study therapist may transfer participants between menu 1 and menu 2 according to their clinical opinion. Participants are given a written do programme to carry out by themselves or with a carer (following preparation) on the weekdays they are not attending therapy.
If the participant is currently receiving rehabilitation, so the upper limb therapy programme is delivered in that setting e.g. stroke unit, out-patients, twenty-four hour period hospital or home. In each case, the study therapist liaises closely with the rehabilitation squad to ensure the participant's needs are addressed and well co-ordinated. At the end of the four week intervention period patients are given advice by the written report therapist regarding maintaining upper limb function.
Participants are reviewed by the research team every three months. If further therapy is required, this is provided past a study therapist. Those in the intervention grouping may also receive a further botulinum toxin type A injection. Participants in both the intervention and control group who have symptomatic spasticity at the 12 month follow up engagement are referred to a spasticity clinic.
Participants who make a good recovery prior to completing the four calendar week upper limb therapy program are discharged from the program provided that they accept a maximum score on the ARAT[20] and have achieved their upper limb goals.
Outcome assessments
Outcomes are measured by an assessor who is blinded to the randomisation grouping one month (+/- 3 days), 3 months (+/- v days) and twelve months (+/- 5 days) afterwards the baseline visit. Before contacting the participant a member of the research team checks with their full general do or stroke unit that they are all the same alive and checks their accost. The medical records of participants who have died are reviewed to seek details of the crusade and circumstances of death, resources utilisation information, and whatsoever potential side effects from botulinum toxin. Each consequence assessment consists of two stages – phase 1 upshot assessment past a self completion postal questionnaire (Barthel ADL Index[26], quality of life (Stroke Impact Scale[27], Euroqol EQ-5D[28], resources utilisation) which is sent to participants ane week prior to stage 2. Participants are asked to bring the completed proforma to their phase 2 appointment.
Stage 2 consequence assessments consists of assessment of upper limb impairment and function (ARAT[20], Motricity Alphabetize[21], grip force[22], Nine Pigsty Peg Exam[23], assessment of spasticity[24] and upper limb hurting[29]) and face to face interview seeking participants experience and views of the study treatment. Data is sought nearly side effects, use of other antispasticity treatment and analgesia for post stroke upper limb pain. Any modify in the participant's concomitant medications since the previous visit is noted. Any new agin events or changes in existing agin events that have occurred since the previous visit are sought. The stage 1 questionnaire is checked for completeness.
Blinding
Outcome assessments are undertaken by an assessor who is blinded to the randomisation grouping. To enable blinding to be achieved study therapists undertake screening and baseline assessments and provide the upper limb therapy programme in one research centre and undertake effect assessments in next centres. At each issue assessment he/she is asked to record if he or she had become unblinded. Participants and the study therapists who provide the upper limb therapy program are not bullheaded to the randomisation group.
Prophylactic evaluation
Side furnishings of botulinum toxin type A are generally mild and transient. Local muscle weakness may occur every bit a result of toxin spread to nearby muscles. 5 per cent experience flu like symptoms 1 week to 10 days post injection, dry out oral cavity and pain at the injection site can occur. Transient dysphagia has been reported. Anaphylaxis rarely occurs. Excessive doses may produce distant and profound neuromuscular paralysis. Respiratory support may be required where excessive doses cause paralysis of respiratory muscles. The safety of botulinum toxin type A in the treatment of participants with upper limb spasticity post stroke is evaluated by examining the occurrence of all adverse and serious adverse events as defined by the Eu clinical trial directive[54]. Follow-upward of each adverse issue continues until the effect or its sequelae resolve or stabilise at a level that is acceptable to the investigator.
Resource utilisation and economical evaluation
Health and social service resource utilise associated with botulinum toxin type A therapy is costed co-ordinate to established methods [55–58]. Measurement and valuation of resources utilize is undertaken using a combination of routine administrative information and master information collection methods devised specifically for the study. Costs measured and valued include the costs of the drugs, costs of the upper limb rehabilitation programme (due east.g. staff time) and costs associated with any adverse side effects associated with the administration of the drugs (e.g. general practitioner or other health care contacts, contacts with social services, out of pocket payments past participants for pain relieving drugs). The costs of the drugs is ascertained from suppliers. Staff fourth dimension is recorded and costed using gross hourly wage rates derived from the mid-point of advisable salary scales. Costs of rehabilitation equipment e.yard. upper limb splinting is obtained from routine data. The frequency of participant contacts with wellness professionals and social services is ascertained through the assistants of a participant questionnaire at the one, 3 and 12 month outcome assessments. Participants are asked about whatsoever contacts they have had with health care professionals and social services since their last assessment and to indicate whatsoever out of pocket expenditure they have incurred for medication. Every bit the study menstruation is 12 months, there is no need to apply discounting. The economic evaluation combines the toll information with information on outcomes from the Euroqol EQ-5D[28] which is used to estimate the quality adjusted life years (QALYS) gained from the intervention. An incremental cost per QALY gained for botulinum toxin plus upper limb therapy relative to therapy solitary will be estimated and associated cost-effectiveness acceptability curves presented. Sensitivity analysis will exist undertaken to assess the sensitivity of the results to variations in key parameters.
Study schedule
Tabular array 1 summarises the study schedule.
Statistical assay
The primary endpoint is the ARAT[20] score at one month. The analysis will be undertaken on an "intention to treat" basis; participants will analysed in the group to which they were randomised.
For each participant information technology volition be determined if there has been a significant comeback in function based on the modify in ARAT score. A successful event volition exist defined equally:
- (i)
a change of 3 or more than points on the ARAT scale for a participant whose baseline ARAT score is between 0 and iii
- (ii)
a change of half-dozen or more points on the ARAT scale for a participant whose baseline ARAT score is between 4 and 51
- (3)
a final ARAT score of 57 for a participant whose baseline ARAT score is 52 or greater.
The proportion of "successes" in each grouping volition and so exist compared using Fisher's exact test. An interval guess of the relative odds of a successful outcome in each group will also be calculated. This interval judge of effect size will be taken forward into the economic evaluation.
Secondary outcomes will as well be analysed on an "intention to care for" basis. A exam appropriate to the blazon of variable will exist undertaken.
A pre-specified sub group analysis will consider the effect of time since stroke as a covariate. It is hypothesised that participants who accept recently had a stroke will have a improve response to handling than those who had a stroke some time ago.
The power calculation used prognosis based methodology[59]. A clinically of import treatment effect is defined every bit a difference in proficient outcomes between intervention and control groups of 15% where a practiced outcome is defined above for each of the ARAT group; information technology is expected to see xx% of the control group accomplish expert outcomes and 35% of the intervention group achieve good outcomes. Using Fleiss' method[60] for a binary outcome and inflating the sample size past 10% to allow for attrition we need to recruit a total sample of 332 participants to give us fourscore% power to discover a fifteen% divergence in good outcomes assuming a two-tailed test and a significance level of 5%. We are aiming to recruit l% of the sample from the ARAT 0–three group and 50% from the ARAT 4–56 grouping.
Current written report status
The study commenced recruitment in July 2005 and achieved target recruitment in March 2008. Participant follow up was completed in June 2008. The results of the study will be submitted for publication end 2008.
Amendments to the report since commencement (July 2005)
(1) Objectives
The study protocol includes measurement of spasticity at the elbow past a biomechanical device which has been used in a previous airplane pilot study[61]. This was to be used in improver to clinical measures. Unfortunately the device is not at a stage of development where it can exist used in a multicentre study.
(two) Setting
Initially the trial was planned in 4 geographical areas: N Tyneside, Wansbeck, Newcastle and Sunderland. Due to depression recruitment rates, further sites were added.
(3) Instance observation
This was widened to include identification of participants from stroke clubs and day centres (in addition to clinical settings) due to initial depression recruitment rates.
(4) Inclusion criteria
Prospective studies of upper limb recovery accept shown that baseline impairment is a potent predictor of outcome. To demonstrate whether botulinum toxin plus upper limb therapy can improve arm office (master outcome), it was initially thought of import to exclude those participants with no retained upper limb function (ARAT 0–3). Due to initial low recruitment, this was later reconsidered and information technology was decided that it would be valuable to include stroke patients with all levels of reduced upper limb function (ARAT 0–56).
The study as well initially excluded participants with cognitive impairment or pregnant speech bug measured past the Abbreviated Mental Test Score and Sheffield Aphasia Screening Test. This was felt to exist also restrictive, excluding patients who were neat to participate. This inclusion criteria was relaxed to include all participants capable of performing the ARAT and complying with the upper limb therapy programme.
(v) Upper limb therapy programme
A 2nd menu was developed for the upper limb therapy programme subsequently the eligibility criteria were widened to include participants with no active upper limb function. This alternative menu was designed because the original menu contained activities which these participants would not have been able to undertake.
(vi) Statistical analysis
Inclusion of participants with lower ARAT scores required revision of the primary analysis and power calculation. Expert opinion concluded that participants with a baseline ARAT of 0–iii could not be predicted to improve as much as those with a baseline ARAT of 4–56. This led to the definition of successful handling equally improvement by iii points on the ARAT for those with a starting ARAT of 0–iii and half-dozen points past those with a starting ARAT of iv–56. Comparison of proportions of successes between the groups (control/intervention) became the chief analysis (as opposed to comparison of absolute ARAT scores in the initial protocol).
The power calculation was revised for the new binary outcome.
(seven) Follow up period
Participants recruited later on 2nd July 2007 were followed for 3 months only. This was a pragmatic determination taken due to the trial being behind schedule from initial low recruitment rates. Curtailing 12 calendar month follow up allows the trial to be completed within the initial study timetable. Twelve month follow up has occurred for 208/333 (62%) participants.
Study acronym
BoTULS: Botulinum Toxin for the Upper Limb later on Stroke
Ethical arrangements and research governance
The written report has MREC, MHRA and local site R+D approvals, and is being conducted in accordance with ICH-GCP[62], the UK Medicines for Human Use (Clinical Trials) Regulations 2004[54] and the Enquiry Governance Framework for Health and Social Care[63].
The study has a Data Monitoring and Ethics Committee (Professor Martin Dennis (chair), University of Edinburgh; Professor Ian Ford, University of Glasgow; Professor Marion Walker, University of Nottingham; Dr Nick Steen, Newcastle University) and a Trial Steering Committee (Professor Michael Barnes, Newcastle Academy; Professor Bipin Bhakta, University of Leeds; Mrs Beryl Fairless, lay fellow member, Newcastle upon Tyne; Professor Gary Ford, Newcastle University; Professor Anne Forster, University of Leeds; Professor Peter Langhorne (chair), University of Glasgow; Professor Helen Rodgers, Newcastle University).
The report sponsor is Newcastle Upon Tyne NHS Foundation Trust.
The study is adopted past the UK Stroke Research Network.
References
-
Skilbeck CE, Wade DT, Langton-Hewer R, Wood VA: Recovery after stroke. Periodical of Neurology, Neurosurgery and Psychiatry. 1983, 46: v-eight. 10.1136/jnnp.46.1.5.
-
Heller A, Wade DT, Wood VA, Sunderland A, Langton-Hewer R, Ward Due east: Arm function afterwards stroke: measurement and recovery over the kickoff 3 months. J Neurol Neurosurg Psychiatry. 1987, 50 (6): 714-719. 10.1136/jnnp.50.6.714.
-
Pandyan AD, Gregoric M, Barnes MP, Wood D, Van Wijck F, Burridge J, Hermens H, Johnson GR: Spasticity: clinical perceptions, neurological realities and meaningful measurement. Disability & Rehabilitation. 2005, 27: two-six. x.1080/09638280400014576.
-
Lance JW: Symposium Synopsis. Spasticity: Disordered Motor Control. Edited past: Feldman RG, Young RR, Koella WP. 1980, Chicago, London: Symposium Specialists Inc, Year Volume Medical Publishers
-
Royal College of Physicians: Guidelines for the use of botulinum toxin (BTX) in the direction of spasticity in adults. 2002
-
Barnes M, Bhakta B, Moore P, Richardson D, Salisbury C, Thornton H, Turner-Stokes L, Ward A: The management of adults with spasticity using botulinum toxin: a guide to clinical practice. Radius Healthcare, Surrey. 2001
-
Simpson DM, Alexander DN, O'Brien CF, Tagliati Thousand, Aswad Equally, Leon JM, Gibson J, Mordaunt JM, Monaghan EP: Botulinum toxin type A in the treatment of upper extremity spasticity: a randomized, double-blind, placebo-controlled trial. Neurology. 1996, 46: 1306-1310.
-
Hesse S, Reiter F, Konrad M, Jahnke MT: Botulinum toxin type A and brusk-term electric stimulation in the treatment of upper limb flexor spasticity after stroke: a randomized, double-bullheaded, placebo-controlled trial. Clinical Rehabilitation. 1998, 12: 381-388. 10.1191/026921598668275996.
-
Bakheit AM, Thilmann AF, Ward AB, Poewe Westward, Wissel J, Muller J, Benecke R, Collin C, Muller F, Ward CD, Neumann C: A randomized, double-blind, placebo-controlled, dose-ranging written report to compare the efficacy and safety of three doses of botulinum toxin blazon A (Dysport) with placebo in upper limb spasticity after stroke. Stroke. 2000, 31: 2402-2406.
-
Bhakta BB, Cozens JA, Chamberlain MA, Bamford JM: Impact of botulinum toxin type A on disability and carer burden due to arm spasticity after stroke: a randomised double blind placebo-controlled trial. Journal of Neurology, Neurosurgery and Psychiatry. 2000, 69: 217-221. 10.1136/jnnp.69.2.217.
-
Bakheit AM, Pittock Due south, Moore AP, Wurker M, Otto S, Erbguth F, Coxon Fifty: A randomized, double-blind, placebo-controlled report of the efficacy and prophylactic of botulinum toxin type A in upper limb spasticity in patients with stroke. European Journal of Neurology. 2001, viii: 559-565. x.1046/j.1468-1331.2001.00277.10.
-
Brashear A, Gordon MF, Elovic E, Kassicieh VD, Marciniak C, Exercise Thou, Lee C, Jenkins S, Turkel C: Intramuscular injection of botulinum toxin for the treatment of wrist and finger spasticity after stroke. New England Journal of Medicine. 2002, 347: 395-400. 10.1056/NEJMoa011892.
-
Brashear A, McAfee AL, Kuhn ER, Fyffe J: Botulinum toxin blazon B in upper-limb poststroke spasticity: a double-bullheaded, placebo-controlled trial. Archives of Physical Medicine & Rehabilitation. 2004, 85: 705-709. ten.1016/j.apmr.2003.08.077.
-
Childers MK, Brashear A, Jozefczyk P, Reding K, Alexander D, Good D, Walcott JM, Jenkins SW, Turkel C, Molloy PT: Dose-dependent response to intramuscular botulinum toxin type A for upper-limb spasticity in patients after a stroke. Archives of Physical Medicine & Rehabilitation. 2004, 85: 1063-1069. x.1016/j.apmr.2003.10.015.
-
Suputtitada A, Suwanwela NC: The lowest constructive dose of botulinum A toxin in adult patients with upper limb spasticity. Disability & Rehabilitation. 2005, 27: 176-184. 10.1080/09638280400009360.
-
van Kuijk AA, Geurts AC, Bevaart BJ, van Limbeek J: Treatment of upper extremity spasticity in stroke patients past focal neuronal or neuromuscular blockade: a systematic review of the literature. Journal of Rehabilitation Medicine. 2002, 34: 51-61. x.1080/165019702753557836.
-
Cardoso E, Rodrigues B, Lucena R, Oliveira IRd, Pedreira One thousand, Melo A: Botulinum toxin blazon A for the treatment of the upper limb spasticity after stroke: a meta-analysis. Arquivos de Neuro-Psiquiatria. 2005, 63: 30-33. 10.1590/S0004-282X2005000100006.
-
Garces K, McCormick A, McGahan L, Skidmore B: Botulinum Toxin A for upper and lower limb spasticity: a systematic review [Technology report no 51]. 2005, Ottawa: Canadian Analogous Part for Health Technology Cess
-
Stroke Unit Trialists Collaboration: Organised inpatient (stroke unit) care for stroke. Cochrane Database Syst Rev. 2007, CD000197-4
-
Lyle RC: A operation test for cess of upper limb office in concrete rehabilitation handling and research. International Journal of Rehabilitation Research. 1981, 4: 483-492. x.1097/00004356-198112000-00001.
-
Demeurisse Thousand, Demol O, Robaye Eastward: Motor evaluation in vascular hemiplegia. European Neurology. 1980, 19: 382-389. 10.1159/000115178.
-
Sunderland A, Tinson D, Bradley L, Hewer RL: Arm function later on stroke. An evaluation of grip strength equally a measure of recovery and a prognostic indicator. J Neurol Neurosurg Psychiatry. 1989, 52 (11): 1267-1272. 10.1136/jnnp.52.11.1267.
-
Sharpless JW: The ix hole peg test of finger hand coordination for the hemiplegic patient. Mossman'due south A Trouble Orientated Arroyo to Stroke Rehabilitation. 1982, Illinois: Charles C Thomas, 2nd
-
Bohannon RW, Smith MB: Interrater reliability of a modified Ashworth scale of muscle spasticity. Physical Therapy. 1987, 67: 206-207.
-
Police M, Baptiste Due south, McColl M, Opzoomer A, Polatajko H, Pollock Due north: The Canadian Occupational Operation Mensurate: An Result Measure for Occupational Therapy. Canadian Journal of Occupational Therapy – Revue Canadienne d Ergotherapie. 1990, 57: 82-87.
-
Mahoney FI, Barthel DW: Functional Evaluation: the Barthel Index. Maryland State Medical Journal. 1965, xiv: 61-65.
-
Duncan PW, Bode RK, Min Lai S, Perera S: Rasch analysis of a new stroke-specific outcome scale: the Stroke Touch Scale. Archives of Physical Medicine & Rehabilitation. 2003, 84: 950-963. x.1016/S0003-9993(03)00035-2.
-
Euroquol Group: Euroquol – a new facility for the measurement of health-related quality of life. Health Policy. 1990, 16: 199-290. 10.1016/0168-8510(xc)90421-ix.
-
Price DD, Bush FM, Long S, Harkins SW: A comparison of pain measurement characteristics of mechanical visual analogue and unproblematic numerical rating scales. Pain. 1994, 56: 217-226. 10.1016/0304-3959(94)90097-3.
-
Hodkinson HM: Evaluation of a mental test score for assessment of mental impairment in the elderly. Age and Ageing. 1972, 1: 233-239. 10.1093/ageing/1.4.233.
-
Al-Khawaja I, Wade DT, Collin CF: Bedside screening for aphasia: a comparing of two methods. Journal of Neurology. 1996, 243: 201-204. 10.1007/BF02444015.
-
Bamford JL, Sandercock PAG, Warlow CP, Slattery J: Inter-observer understanding for the assessment of handicap in stroke patients. Stroke. 1989, twenty: 828-844.
-
Bamford J, Sandercock P, Dennis M, Burn down J, Warlow C: Nomenclature and natural history of clinically identifiable subtypes of cerebral infarction. Lancet. 1991, 337: 1521-1526. 10.1016/0140-6736(91)93206-O.
-
Royal College of Physicians: National clinical guidelines for stroke (2nd edition). 2004
-
Wade D: Goal planning in stroke rehabilitation: evidence. Topics in Stroke Rehabilitation. 1999, 6: 37-42. 10.1310/FMYJ-RKG1-YANB-WXRH.
-
Pomeroy Five, Tallis R: Restoring move and functional power subsequently stroke: now and the futurity. Physiotherapy. 2002, 88: three-17. x.1016/S0031-9406(05)60524-X.
-
Lee van der JH, Snels IA, Beckerman H, Lankhorst GJ, Wagenaar RC, Bouter LM: Exercise therapy for arm function in stroke patients: a systematic review of randomized controlled trials. Clinical Rehabilitation. 2001, 15: 20-31. x.1191/026921501677557755.
-
Kwakkel Thousand, Wagenaar R, Twisk J, Lankhorst G, Koetsier J: Intensity of leg and arm grooming after primary heart-cognitive-artery stroke: a randomised trial. Lancet. 1999, 354: 191-196. x.1016/S0140-6736(98)09477-X.
-
Dean CM, Richards CL, Malouin F: Chore-related circuit training improves functioning of locomotor tasks in chronic stroke: a randomized, controlled pilot trial. Archives of Concrete Medicine & Rehabilitation. 2000, 81: 409-417. 10.1053/mr.2000.3839.
-
Dean CM, Shepherd RB: Task-related training improves performance of seated reaching tasks after stroke. A randomized controlled trial. Stroke. 1997, 28: 722-728.
-
Carr J, Shepherd RB: Stroke Rehabilitation. Guidelines for exercise preparation to optimise motor skill. 2003, London: Butterworth Heinemann
-
Barnes MP, Johnson GR: Upper Motor Neurone Syndrome and Spasticity. 2001, Cambridge: Cambridge University Press, First
-
Barnes Chiliad, Dobkin B, Bogousslavsky J: Recovery after stroke. 2005, Cambridge: Cambridge University Press
-
Edwards S: Neurological Physiotherapy. 2002, Edinburgh: Churchill Livingstone, two
-
Lannin NA, Herbert RD: Is mitt splinting effective for adults post-obit stroke? A systematic review and methodologic critique of published research. Clinical Rehabilitation. 2003, 17: 807-816. x.1191/0269215503cr682oa.
-
Guidelines on Stroke. http://www.sign.air conditioning.uk
-
Stokes M: Concrete Direction in Neurological Rehabilitation. 2005, Edinburgh: Elsevier Mosby, 2
-
Van Peppen RPS, Kwakkel Grand, Wood-Dauphinee S, Hendriks HJM, Wees Van der PJ, Dekker J: The impact of physical therapy on functional outcomes after stroke: what's the prove?. Clinical Rehabilitation. 2004, 18: 833-862. 10.1191/0269215504cr843oa.
-
Hanlon RE: Motor learning following unilateral stroke. Archives of Physical Medicine & Rehabilitation. 1996, 77: 811-815. 10.1016/S0003-9993(96)90262-two.
-
Magill RA: Motor Learning: Concepts and Applications. 2001, Boston: McGraw Hill
-
Shea JB, Morgan RL: Contextual interference furnishings on the acquisition, retention and transfer of a motor skill. Journal of Experimental Psychology: Learning, Memory and Knowledge. 1979, 5: 179-187.
-
Duncan Pow: Synthesis of intervention trials to ameliorate motor recovery post-obit stroke. Topics in Stroke Rehabilitation. 1997, 3:
-
van Wijck F, Mackenzie J, Hooper J, Barnes Yard, Johnson Thousand: Improving arm role in patients with chronic spasticity: combining botulinum toxin with a task orientated motor learning programme. International Journal of Rehabilitation Research. 2004, 27 (suppl 1): 109-
-
Medicines for Man Utilize (Clinical Trials) Regulations 2004. http://www.uk-legislation.hmso.gov.uk/si/si2004/20041031.htm
-
Donaldson C, Shackley P: Economic science evaluation. 1997, Oxford: Oxford Academy Press, 3
-
Drummond MF, O'Brien B, Stoddart GL, Torrance GW: Methods for the economical evaluation of health care. 1997, Oxford: Oxford University Press, 2
-
Golden M, Siegel J, Russell L, Weinstein M: Price effectiveness in health and medicine. 1996, New York: Oxford Academy Press
-
McNamee P, Christensen J, Soutter J, Rodgers H, Craig N, Pearson P, Bail J: Cost analysis of early supported infirmary discharge for stroke. Age and Ageing. 1998, 345-351. 10.1093/ageing/27.3.345.
-
Mendelow Advert, Gregson BA, Fernandes HM, Murray GD, Teasdale GM, Hope DT, Karimi A, Shaw MDM, Barer DH: Early on surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. Lancet. 2005, 365: 387-397.
-
Fleiss JL: Statistical Methods for Rates and Proportions. 1981, New York: Wiley
-
Pandyan Advertisement, Vuadens P, van Wijck FMJ, Stark South, Johnson GR, Barnes MP: Are nosotros underestimating the clinical efficacy of botulinum toxin (type A)? Quantifying changes in spasticity, strength and upper limb function afterwards injections of Botox to the elbow flexors in a unilateral stroke population. Clinical Rehabilitation. 2002, sixteen: 654-660. 10.1191/0269215502cr536oa.
-
ICH Harmonised Tripartite Guideline for Good Clinical Exercise. http://www.ich.org
-
Department of Health: Research Governance Framework for Wellness and Social Care. 2005, 2nd
Acknowledgements
Nosotros would like to admit the contribution of Dr Tony Field, Consultant in Rehabilitation Medicine, Hunters Moor Regional Rehabilitation Centre, Newcastle who was involved in designing and planning the projection. He sadly died in 2005.
We would especially similar to give thanks the following for their contribution:
(1) Patients who are participating in the trial.
(two) Written report therapists: Lydia Aird, Louise Baxter, Lianne Brkic, Caroline Deacon, Hilary Ford, Heather Hunter, Nina Lishman, Paul McNeillie, Dadirayi Mhiripiri, Janet Nesbitt, Frances Sapsford.
(3) Co-ordinating centre squad: Jessie Cowe, Katharine Forster, Linda Goulbourne, Anne Harrison, Joseph Hoben, Debbie Jones, Ruth Forest.
(4) Primary investigators: Prof David Barer (Gateshead Health NHS Trust), Dr David Bruce (North Tees and Hartlepool NHS Trust), Dr Tim Cassidy (City Hospitals Sunderland NHS Foundation Trust and Gateshead Health NHS Trust), Dr Paul Davies (North Cumbria Acute Hospitals NHS Trust), Dr Phil Earnshaw (County Durham and Darlington NHS Foundation Trust), Dr Akif Gani (Newcastle Primary Care Trust), Prof Chris Gray(City Hospitals Sunderland NHS Foundation Trust), Dr Ali Mehrzard (County Durham and Darlington NHS Foundation Trust), Dr Jon Scott (S Tyneside NHS Foundation Trust).
(5) Neuro-outpatient staff at the International Centre for Neurorehabilitation, Newcastle upon Tyne who assisted with botulinum clinics.
(6) Sandra Stark and Julia Johnson (International Middle for Neurorehabilitation, Newcastle upon Tyne), who assisted with the upper limb therapy programme design.
(7) DMEC external members: Prof Martin Dennis, (chair) (Academy of Edinburgh), Prof Ian Ford (University of Glasgow), Prof Marion Walker (University of Nottingham).
(8) TSC external members: Prof Peter Langhorne (chair) (University of Glasgow), Prof Bipin Bhakta (University of Leeds), Mrs Beryl Fairless (lay member), Prof Anne Forster (University of Leeds).
(9) Ipsen Ltd who provided advice, back up and the botulinum toxin type A (Dysport®).
(x) United kingdom of great britain and northern ireland Stroke Research Network: Karin Bail, Ellen Brown, Claire Garner, Penny Williams.
We would too like to thank clinical services, Newcastle Academy, NHS R&D and human resources departments in participating centres that enabled the study to be undertaken.
The study is funded by the NIHR Health Technology Assessment Programme. The views and opinions expressed here are those of the authors and practise not necessarily reflect those of the Department of Health.
LS is funded from the grant. NS receives payment from the grant. The NIHR HTA Program provided the research question and was involved in designing and approving the trial protocol.
Author data
Affiliations
Consortia
BoTULS investigators
Corresponding author
Additional information
Competing interests
Ipsen Ltd are providing the botulinum toxin type A used past the written report free of accuse. They have also provided funding for sandwich lunches at launch meetings at written report sites. The pattern, analysis and reporting of the study is existence undertaken independently of Ipsen Ltd. Hour is the deputy director of the Great britain Stroke Research Network. LS has no competing interests. CP is the deputy managing director of the N East Stroke Inquiry Network. FvW has no competing interests. MB is a consultant in rehabilitation medicine and uses botulinum toxin regularly in clinical practice. He has received sponsorship from Ipsen Ltd to attend and teach at conferences and meetings merely has no personal financial interest in botulinum toxin or any related product. LG is a consultant in rehabilitation medicine and uses botulinum toxin regularly in clinical do. She has received sponsorship from Ipsen Ltd to nourish and teach at conferences and meetings only has no personal financial involvement in botulinum toxin or whatsoever related product. GF is director of the U.k. Stroke Research Network. PS has no competing interests. NS has no competing interests.
Authors' contributions
Hour, CP, FvW, MB, GF designed the RCT. HR is the chief investigator. 60 minutes and LS drafted the newspaper. LS and CP undertook the literature review of previous trials. LS is the clinical research associate with responsibility for the 24-hour interval to day running of the written report and delivery of botulinum toxin type A injections. FvW designed the upper limb therapy program. She trained the research therapists to evangelize the programme and administer the upper limb outcome measures. MB and LG provide expertise in the direction of spasticity and use of botulinum toxin. LG has as well been involved in the delivery of trial botulinum toxin blazon A injections. PS designed and is responsible for the health economic science component of the study. NS is the report statistician. All authors take commented upon drafts of the newspaper and accept given final blessing to this version.
Authors' original submitted files for images
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Admission article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/two.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original piece of work is properly cited.
Reprints and Permissions
About this article
Cite this article
Rodgers, H., Shaw, 50., Cost, C. et al. Study blueprint and methods of the BoTULS trial: a randomised controlled trial to evaluate the clinical effect and cost effectiveness of treating upper limb spasticity due to stroke with botulinum toxin type A. Trials 9, 59 (2008). https://doi.org/10.1186/1745-6215-ix-59
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/ten.1186/1745-6215-9-59
Keywords
- Botulinum Toxin
- Stroke Unit
- Botulinum Toxin Type
- Limb Function
- Dysport
Source: https://trialsjournal.biomedcentral.com/articles/10.1186/1745-6215-9-59
0 Response to "Botulinum Neurotoxins for Postã¢â‚¬âstroke Spasticity in Adults a Systematic Review"
Post a Comment